Can we capture
methane from the Arctic seabed?
Stephen H. Salter, School of Engineering, University of Edinburgh,
Scotland.
Prepared
for the John Nissen Methane Workshop, Chiswick 15,16 October 2011.
DRAFT 3 November with pressure ridge
addition.
Methane is a greenhouse gas more than 100
times more effective than carbon dioxide in the short term. It is stored in the form of clathrates which
are unstable if pressure is lower or temperature is higher than a line on a
pressure versus temperature graph. Figure 1 shows that the slope of the
atmospheric concentration has sharply increased since 2007. Previous high levels of methane were
associated with the Permian mass extinction, 250 million years ago.
Figure 1. Anomalies of CH4 mean volume mixing ratios
for Northern and Southern hemispheres courtesy Leonid Yurganov. Updated
mixing ratios (Dlugokencky et al., 2009) were subtracted from the seasonal
cycles averaged over 2003-2007. The right scale shows the anomaly of total mass
of CH4 in the tropospheric layer of each hemisphere. The growth has been
continuing in 2010-2011, according to the updated satellite data by Frankenberg
et al. 2011.
This note discusses the design problems of
a system to deploy kilometre-sized areas of plastic film to collect methane
from suitable areas of the sea bed. The
gas can be flared off at sea to convert it to less damaging carbon dioxide or
perhaps, if there are very high flow rates, recovered by a gas carrier and used
ashore.
There seem to be solutions to what appeared
initially to be an insoluble problem.
The difficulties
When John Nissen first raised the problem
of Arctic methane my initial reaction was that capture at the sea bed would be
impossible. But trying to design for the
impossible can be interesting. It seemed
a useful exercise to identify the reasons for impossibility. We can list
difficulties as follows:
- Methane release at very low flow rates over too wide an area.
- Release at very high rates over a small area such as a well
blow-out.
- Rough seas during deployment.
- The presence of obstructions such as wreckage, rock outcrops,
munitions or steep slopes.
- Fast, variable-direction or unpredictable currents.
- Equipment sinking into very soft ooze on the seabed.
- Hydrogen sulphide toxicity.
- Unacceptable biological consequences due to the presence of equipment.
- The need to recover everything at some date in the future.
- The pressure ridges shown by Peter Wadhams at the Chiswick
workshop.
I now believe that despite these problems
methane can be captured in quite large quantities from areas of several square
kilometres of plastic film in a single installation.
The design
The film sheet is packed into a pair of
left and right-handed rubber trough cases [1] and [2] with a rectangular inner
section as shown in figure 2. Each trough case carries two steel cables [3]. The trough cases would be produced by a
continuous moulding/extrusion machine in lengths of several kilometres using
plant similar to that used for electrical cables. The left and right handed
pair are connected at the centre by two thin isthmus strips of material [4] [5]
above and below a rectangular section passage.
The passage contains a rectangular section runner [6] with two blades
[7] [8] which can be pulled through the full length of the extrusion by a steel
cable. [9]. If the steel cable is pulled the two blades
will cut the connection strips and the trough case halves will be
separated.
The underside of the trough case extrusion has a moulded tread
with a pattern of saw-tooth section ridges [10] lying at an angle of about 30
degrees to the length of the extrusion. This ridge angle is an important design
parameter. At the outer corners of the
bottom of the insides of the trough cases are recesses [11] into which a bead
on the edge of an extruded plastic sheet can be pushed. The outer walls of the trough are much
thicker than the inner walls and contain galleries [12] along which methane can
be transported to riser pipes. They connect to the higher points of the
saw-tooth moulding. A high-density filler is added to the rubber to make sure
that it is heavier than cold sea water but not heavier than the ooze on the sea
bed. The outer edges of the extrusion [13] are sloped like the front of a
sledge.
At the bottom of figure 2 the troughs are
shown filled with a zig-zag stack of flexible plastic with a density just
greater than cold sea water and a thickness of about 200 microns. The zig-zag stacks on each side a joined at
the top [14]. The lower edges with a
bead are pushed into the recesses in each trough. This plastic would be produced by a second
extrusion machine consisting of interdigital plates to be described later. If the width of each trough is one metre and
the trough depth is 150 mm there will be space for 750 layers of zig-zag
plastic, giving an extended width of 1.5 kilometres when the zig-zags on the
two sides are unpacked. The stacks of
plastic film can be packed securely by lid flaps [15] retained by a vacuum
maintained through pipes [16].
The length of plastic and rubber would be
wound in a single scroll on the drum of a pipe-laying vessel such as the Stena
Apache. A drum diameter of 35 meters
could take a width of 1.5 kilometres and length of 3 kilometres, giving a capture
area of 4.5 square kilometres.
Figure 2. Empty and filled extruded rubber trough cases with 4 times enlarged views of end and centre.
Deployment.
Survey vessels with side-scan sonar and
methane detection sensors would look for suitable sites with no large obstructions,
suitable current velocities and comfortable methane emission rates.
Small obstructions can be levelled with
robotic sea bed vehicles such as the one described at the 2011 EWTEC
conference.
The pipe-laying vessel would take station
well downstream of the target area and pay out the scrolled material to the sea
bed as if it were oil pipe. The extreme
flexibility of the trough case (relative to 12 inch steel pipe) would allow
wave tolerant J-lay rather than an S-lay release.
Once the full length of the package is on
the sea bed (figure 3) it would be towed along the seabed by ropes attached to
the fore end of the rubber extrusions until it reached a point before the start
of the target area equal to the string length divided by the cosine of the
ridge angle. If possible the tow
direction should be perpendicular to current and swell.
The central cable with knife blades would
be pulled through the rubber extrusion to separate the two troughs.
The vacuum retaining the lid flaps will be
released.
Towing to increase the width of the film
can now begin. Towing from the pipe-laying vessel would mean lifting the
leading edge of the pack and there might be disturbance by waves. It is preferable to use a horizontal force
from a sea bed walking vehicle. There
might sometimes be an advantage in raising and lowering the leading edge in the
way used for aligning carpets. The tow
force would depend on the weight of the package in water and the coefficient of
friction to the sea bed. This is expected to be about 250 kN. This will set the size of the steel cables
embedded in the rubber extrusions which transmit the tow force along the length
of the rubber and the bollard pull of the tow vehicles.
The tow vehicles will keep the tow lines
pointing along the line of the package but the angled ridges would make the two
troughs move apart from each other and so the tow vehicles will take diverging
courses. The layers of plastic film will be pulled away from the zig-zag stack,
as shown in figure 3, with the weight of the retaining lids providing a gentle
resisting force.. GPS systems will be used to keep the advance rate of the tow
vehicles matched.
The small density difference between
plastic and sea water will mean that the drag friction between plastic and sea
bed will be very low with a factor of safety of several hundred relative to the
plastic strength.
The ridges in the rubber extrusion will
leave furrows on the surface of the seabed.
When the furrows are covered by the plastic sheet they will form
passages for the removal of gas through galleries in the outer walls of the
trough.
The outward movement of the trough cases
will build up material from the sea bed at the front of the outer sledge
faces. Water moving through eductor jets
[17] can move some of the sea bed material over the film.
The gas pipe connection from below the film
to the surface will bring its pressure closer to atmospheric. Eventually several bars of water pressure
will clamp the film and trough casings firmly to the sea bed.
Figure 3. Deployment of the film using the side force
from the inclined ridges at the bottom
of the trough cases. Proportions are grossly
distorted.
Tooling
Thermo-plastic films can be made by heating
pellets of the feed stock to their melting point, pumping the liquid material
through fine gaps in an extrusion tool and progressively cooling the downstream
section of the tool to a temperature at which the film can be handled. The
energy requirement is the sum of melting heat and pumping pressure. Much of the heat can be recycled back to the
incoming feed stock. The product is easier to handle if the pumping is in a
downward direction.
The tool will consist of one inner and two
outer stacks of plates each of which consists of two half plates which have
been machined with a zig-zag coolant channel and then riveted and spot-welded
back together as shown in figure 4. The
key problem is maintaining an accurate gap, probably 200 microns, between inner
and outer plates. Gravitational sag will
be avoided if plates are vertical. At
the top of the tool where the film material is still liquid the gap can be
defined by streamlined shims but in the cooler regions it must be actively
controlled with no physical blockage.
Material from a rolling mill usually has
quite large flatness errors and a skin under compression. The first step will be stress relief by
raising the plate temperature to 650 C for an hour and cooling it slowly.
Toolroom surface grinders can work to a
flatness better than 3 microns but if curved parts are held flat on a magnetic
chuck the curvature will be restored when the magnetic flux is removed. It will be necessary to hold the plates on a
hot wax chuck as used in the optical industry. It might be useful to consider a low-force
cutting technique such as spark erosion.
Figure 4. A grossly distorted plan view of the topology
of the extrusion tool with exploded parts. A 1500 metre width would require 750
plates rather than eight. Maintaining a
gap for the film thickness is a challenging problem but may be done with
differential temperature control. The tool for a 1500 metre width of film would
weigh about 200 tonnes. If the
differential temperature idea is not feasible, smaller tools could be used but
a way to store and join kilometre lengths edge to edge would be needed.
Temporary coiling looks difficult.
Gap control
We can use an array of capacitance
transducers to measure the gap between plates of an assembled stack. We can cover the surfaces of plates with
resistive heating elements either side of the cooling channels. By differential control of the heating
currents we can control the local curvature of a plate. The coefficient of thermal expansion of
stainless steel is 17 part per million per C degree. A temperature difference of 1C across a 15 mm
plate will induce a radius of curvature of 440 metres. If the width of the heating element is 100 mm
this means a deflection of 11 microns.
A neat way to provide plate deflection
control is to divide the plate surfaces into 100 mm squares with a resistive
layer filling most of the area. The
squares would be connected in series and driven with a constant current from a
high impedance source rather than a constant voltage. The current would be diverted around the
heating element by a parallel, high-frequency switch operated for a variable
fraction of the time. A small fraction
of the surface with a grounded guard backing would be given a high-frequency
excitation to measure the capacitance to the adjacent plate.
Cold heat exchanger fluid will be pumped
into the bottom of the vertical tooling plates and emerge from the top at nearly
the melting temperature of the plastic film.
After some extra heating the fluid will then move downwards through a
vertical-tube heat-exchanger to melt the incoming plastic.
Solidified film coming out of the bottom of
the tool will be further cooled by an upward flow air which will then be
directed down through a bed of rising feed pellets and shredded plastic being
recycled. Air can flow easily through
gaps between pellets or shredded feed stock.
The surface area of pellets is large even if heat transfer per unit area
is low. Heat can flow more easily between liquids. However there will be an awkward gap between
solid but nearly molten pellets in the air in the pellet heat exchanger and
liquid in the one above it. Although the
temperature difference might be quite small the amount of latent heat of fusion
might be substantial.
Gas flow rates
Size of release plumes
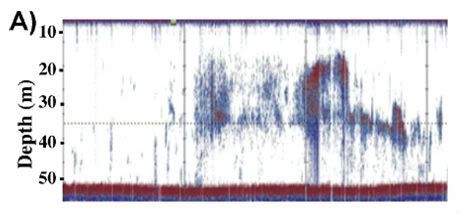
Figure 5. An echo sounder image giving the size of
methane plumes from Shakhova et al.. This shows a transect of about 500 metres
in the Laptev sea showing bubble plume return
features
and also zooplankton other non-bubble scatterers such
as fish.
Material quantities
The Shakhova presentation also mentioned total
areas of methane hot spots of 210,000 square kilometres, the area of a square
of side 460 kilometres. The proposed design
needs about 200 tonnes of plastic film per square kilometre. Total world
consumption of plastics in 2010 was about 300 million tonnes and forecast to
rise to 538 million in 2020. Protecting
the Shakova area with coverings which lasted 10 years would take about 1.5% of
total present world plastic production.
Recovery
Maintenance would be very difficult and is
not planned. But anyone putting anything into the sea has an ethical duty to
plan for its recovery. The proposal is
to make structures of two cutting discs about 2 metres in diameter separated at
12 metres which can roll along the length of the film to cut it into 12 metre
wide strips. The ends of the cut can be
gripped with a vacuum plate, lifted to the surface and wound round a drum. The area of the long side of a 3 km length
sheet of clean film is only 0.6 square metres. Over a period of years it will probably have
acquired biological growths, some of which can be removed by pulling it between
contra-rotating brushes. It is desirable
that growth thickness can be reduced to the level at which film can be packed
into 2.2 metre diameter for movement in a sea container. For a film length of 3 kilometres this means
a thickness of film plus growth of 1.25 mm.
The extruded rubber trough cases would be wound on the drum of a
pipe-laying vessel.
Conclusions.
There is a wide range of estimates for the
rates of methane release from Arctic seabeds but the higher ones are alarming
enough for all defensive measures to be carefully examined.
Initial design work for the manufacture and
deployment of kilometre-sized areas of plastic film to capture methane suggests
that that this may be possible for a range of emission rates provided that the
areas of the sea bed are clear of obstructions. This conclusion should be checked
with people from the plastic and rubber industries.
Deployment and recovery will require
pipe-laying vessels from the oil industry , such as the Stena Apache, and
specialised seabed crawlers which have been designed for wave and tidal-stream
installation.
Unless methane emission rates are even
higher than suggested it will not be economical to recover methane for use on
land and so flaring off at sea is more likely.
However there may be enough energy to drive ice-cutting equipment to
keep the water round a flare stack clear of drifting ice in winter.
The extrusion tool for a 1500 metre width
will require about 200 tonnes of very flat stainless steel sheet. The critical
problem is maintaining an accurate gap in the extrusion tool. This can be done with differential
temperature control of opposite surfaces of a stack of interdigital plates with
central cooling channels.
The separation of halves of a film package
can be done by the force generated from angled saw-tooth ridges on the
underside when the package is dragged over the sea bed. This allows very wide film coverage from an
easily transported package and leaves tracks for methane flow.
If the underside of the film has a pipe
connection to the atmosphere the pressure from water above it will clamp it
firmly to the sea bed.
Work on long-term biological testing of
candidate film materials should begin as soon as possible.
It is necessary to have credible techniques
to recover all materials from the sea bed.
The proposed method must be critically checked by experienced offshore
engineers.
A 4.5 square kilometre area of 200 micron
sheet will need about 930 tonnes or 25 railway trucks of plastic but this is
small compared with world production.
Energy consumption in the present plastics industry is about 10 MJ a
kilogram compared with 2.25 MJ for the latent heat of steam. If the film extrusion velocity is 10 mm a
second we will need 3.5 days for one pack and a power of 35 MW. Heat pump technology could give a very large
reduction in energy consumption and must be carefully investigated.
We may have to avoid deployment in water
depths less than the deepest pressure ridges. The leading ice authority, Peter
Wadhams, says that these can reach down to 34 metres below the surface.
Actions
Resolve the three-order of magnitude dispute
about methane release rates and investigate sea bed methane release rates and their
variability in space and time.
Check design assumptions with the plastic
film and rubber extrusion industry.
Choose the best candidate film materials
with density just greater than cold sea water (1028.4 kg/m3) and establish
stress capability in working conditions.
A large strain length is more important than tensile strength.
Place specimens of the various film types
in suitable test site in northern Norway
Measure tow forces on 5-metre sized blocks
and establish the best ridge angle for a range of sea bed conditions from
gravel to sand to ooze.
Place blocks of various shapes and
densities fitted with accelerometers on the sea bed and measure how many roll
or slide.
Carry out a sonar side-scan survey to
identify obstructions in suitable areas.
Some, such as bullion cargoes, may be removable.
Collect information on depth and occurrence
of pressure ridges in methane release areas.
Pray that the continual underestimation of
the potential climate risks by people who are responsible for defending us
against them does not continue.
Links
World plastic production
Shakhova PowerPoint presentation link.
Shakhova Semiletov paper
Pipe-laying vessels
Other collected papers
References
Dlugokencky, E. J., L. M. P. Bruhwiler, J. W. C. White, L. K. Emmons, P. C. Novelli, S. A. Montzka, K. A. Masarie, P. M. Lang, A. M. Crotwell, J. B. Miller and L. V. Gatti (2009), Observational constraints on recent increases in the atmospheric CH4 burden, Geophysical Research Letters, 36, L18803, 10.1029/2009GL039780.
Frankenberg, C., I. Aben, P. Bergamaschi, E. J. Dlugokencky, R. van Hees, S. Houweling, P. van der Meer, R. Snel P. Dol (2011), Global column-averaged methane mixing ratios from 2003 to 2009 as derived from SCIAMACHY: Trends and variability, Journal of Geophysical Research-Atmospheres, 116(D04302), 1-12, 10.1029/2010JD014849.
Montzka, S. A., E. J. Dlugokencky and J. H. Butler (2011), Non-CO2 greenhouse gases and climate change, NATURE, 476, 43-50, 10.1038/nature10322.
Dlugokencky, E. J., L. M. P. Bruhwiler, J. W. C. White, L. K. Emmons, P. C. Novelli, S. A. Montzka, K. A. Masarie, P. M. Lang, A. M. Crotwell, J. B. Miller and L. V. Gatti (2009), Observational constraints on recent increases in the atmospheric CH4 burden, Geophysical Research Letters, 36, L18803, 10.1029/2009GL039780.
Frankenberg, C., I. Aben, P. Bergamaschi, E. J. Dlugokencky, R. van Hees, S. Houweling, P. van der Meer, R. Snel P. Dol (2011), Global column-averaged methane mixing ratios from 2003 to 2009 as derived from SCIAMACHY: Trends and variability, Journal of Geophysical Research-Atmospheres, 116(D04302), 1-12, 10.1029/2010JD014849.
Montzka, S. A., E. J. Dlugokencky and J. H. Butler (2011), Non-CO2 greenhouse gases and climate change, NATURE, 476, 43-50, 10.1038/nature10322.






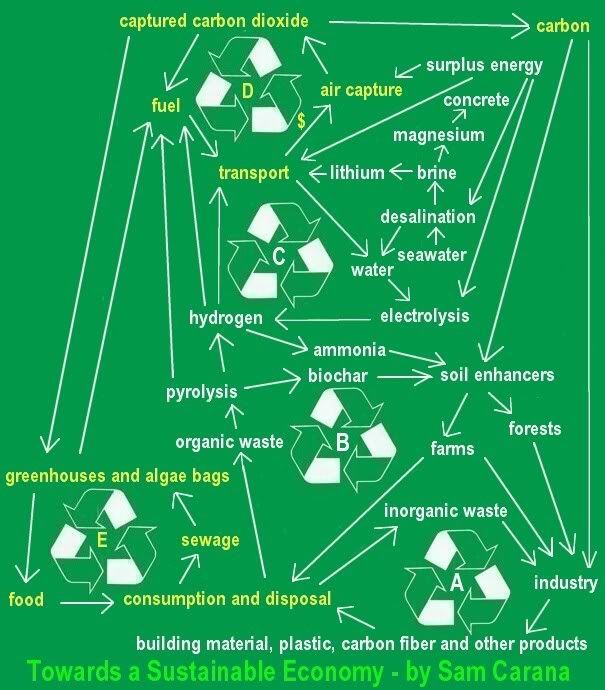
 AIR CAPTURE of CO2 (carbon dioxide) is an essential part of the blueprint to reduce carbon dioxide to acceptable levels. Fees on
AIR CAPTURE of CO2 (carbon dioxide) is an essential part of the blueprint to reduce carbon dioxide to acceptable levels. Fees on  conventional jet fuel seems the most appropriate way to raise funding to help with the development of air capture technology.
conventional jet fuel seems the most appropriate way to raise funding to help with the development of air capture technology.
 Apart from growing algae in greenhouses, we should also consider growing them in bags. NASA scientists
Apart from growing algae in greenhouses, we should also consider growing them in bags. NASA scientists 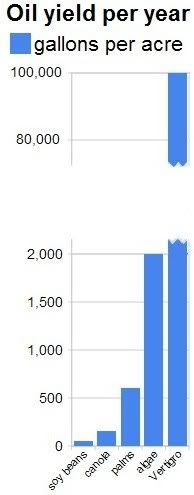 The NASA article conservatively mentions that some types of algae can produce over 2,000 gallons of oil per acre per year. In fact, most of the oil we are now getting out of the ground comes from algae that lived millions of years ago. Algae still are the best source of oil we know.
The NASA article conservatively mentions that some types of algae can produce over 2,000 gallons of oil per acre per year. In fact, most of the oil we are now getting out of the ground comes from algae that lived millions of years ago. Algae still are the best source of oil we know.
 A 2007
A 2007  Professor David Keith (left) of the University of Calgary is working on a tower, 4 feet wide and 20 feet tall, with a fan at the bottom that sucks air in. The tower looks like it's made mainly of plastic, which could be made with carbon produced by such a tower. Inside the tower, limestone or a similar agent is used to bind the CO2 and to split CO2 off by heating it up. The limestone is recycled within the tower, although it does need to be resupplied at some stage. Anyway, the main cost appears to be the electricity to run it.
Professor David Keith (left) of the University of Calgary is working on a tower, 4 feet wide and 20 feet tall, with a fan at the bottom that sucks air in. The tower looks like it's made mainly of plastic, which could be made with carbon produced by such a tower. Inside the tower, limestone or a similar agent is used to bind the CO2 and to split CO2 off by heating it up. The limestone is recycled within the tower, although it does need to be resupplied at some stage. Anyway, the main cost appears to be the electricity to run it.